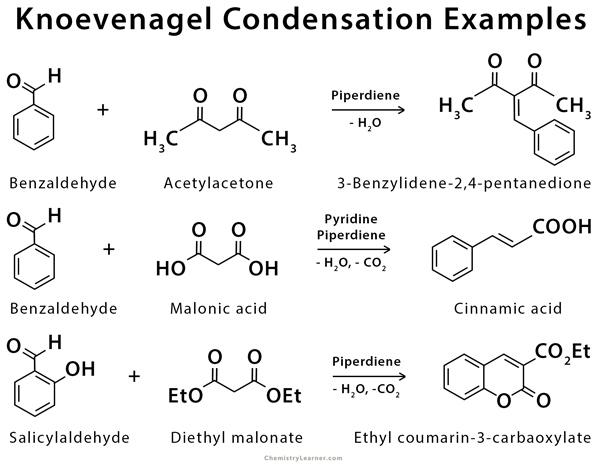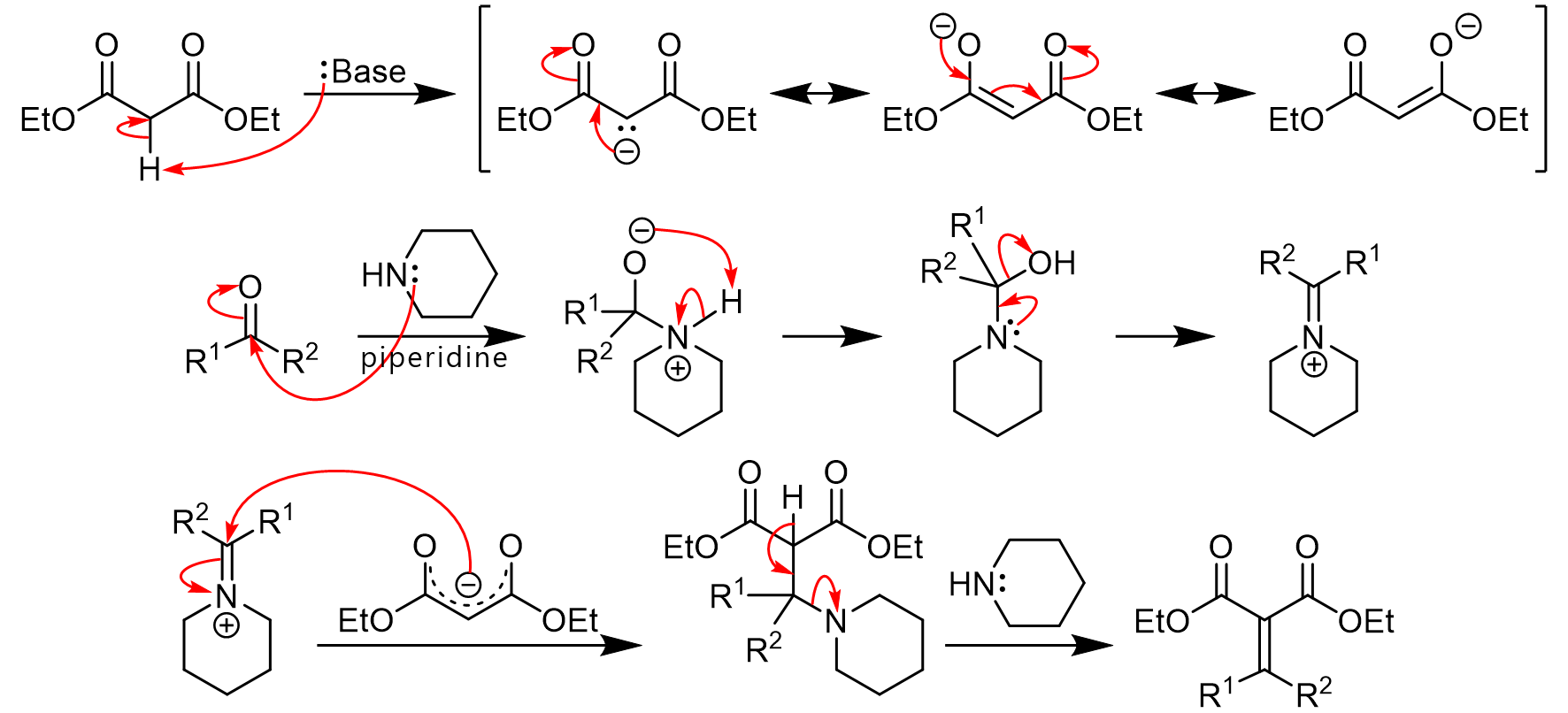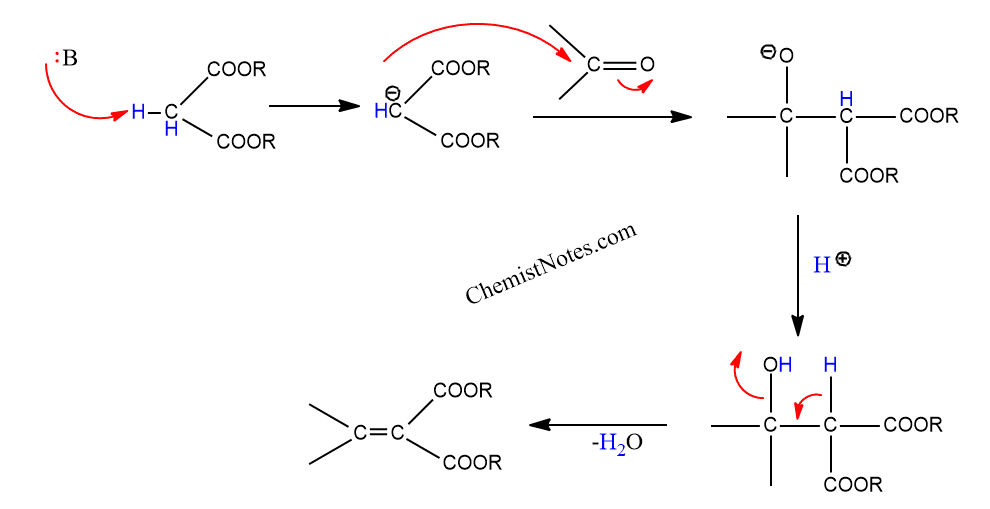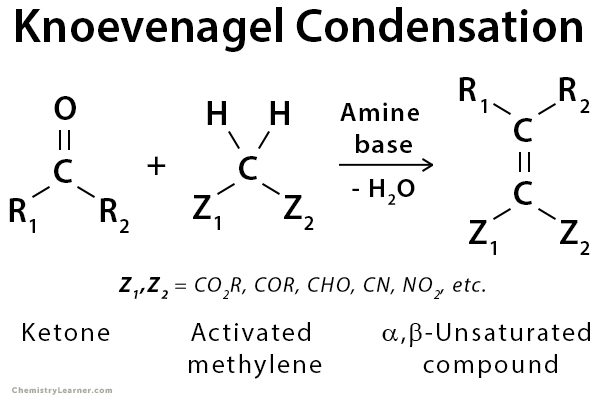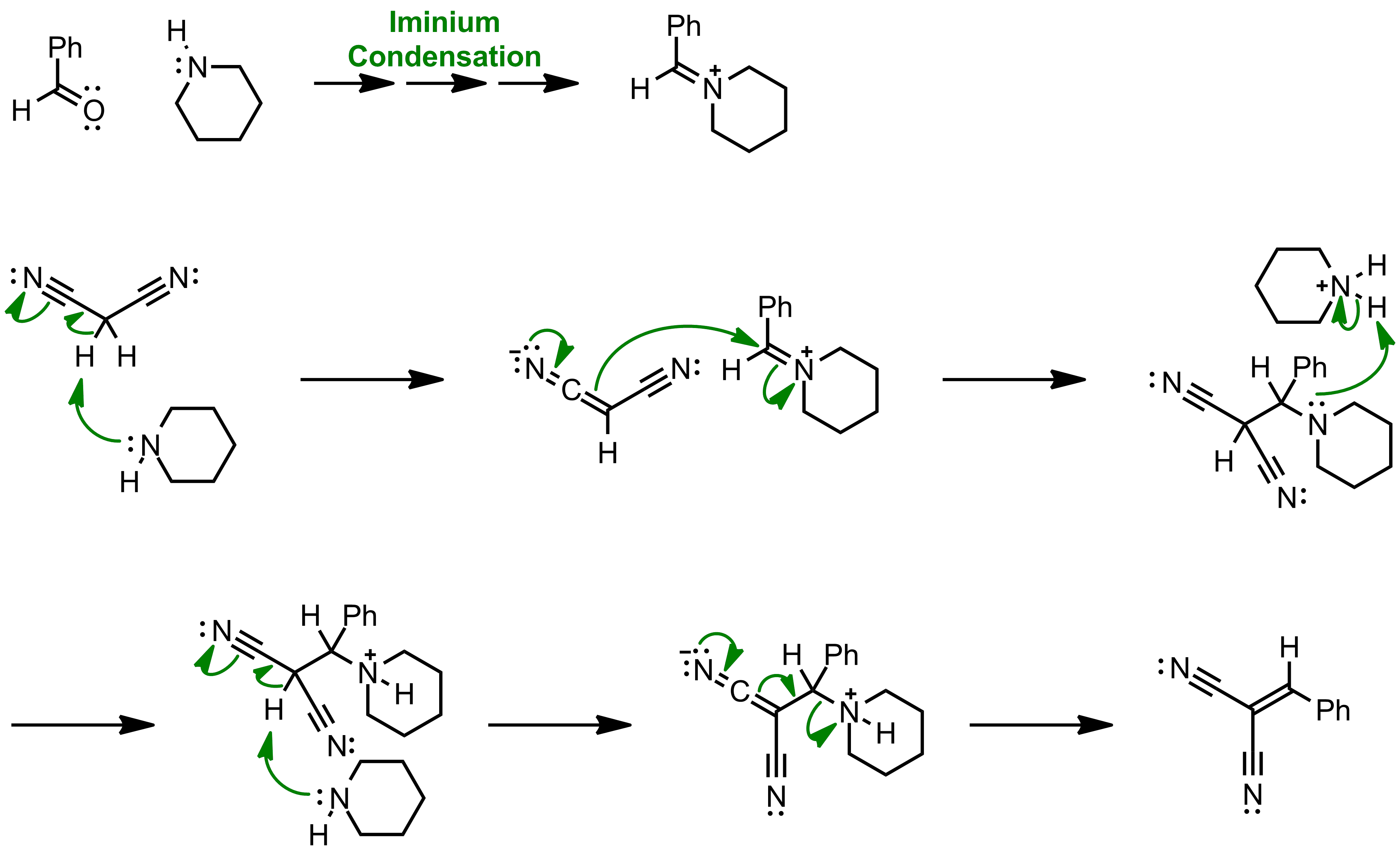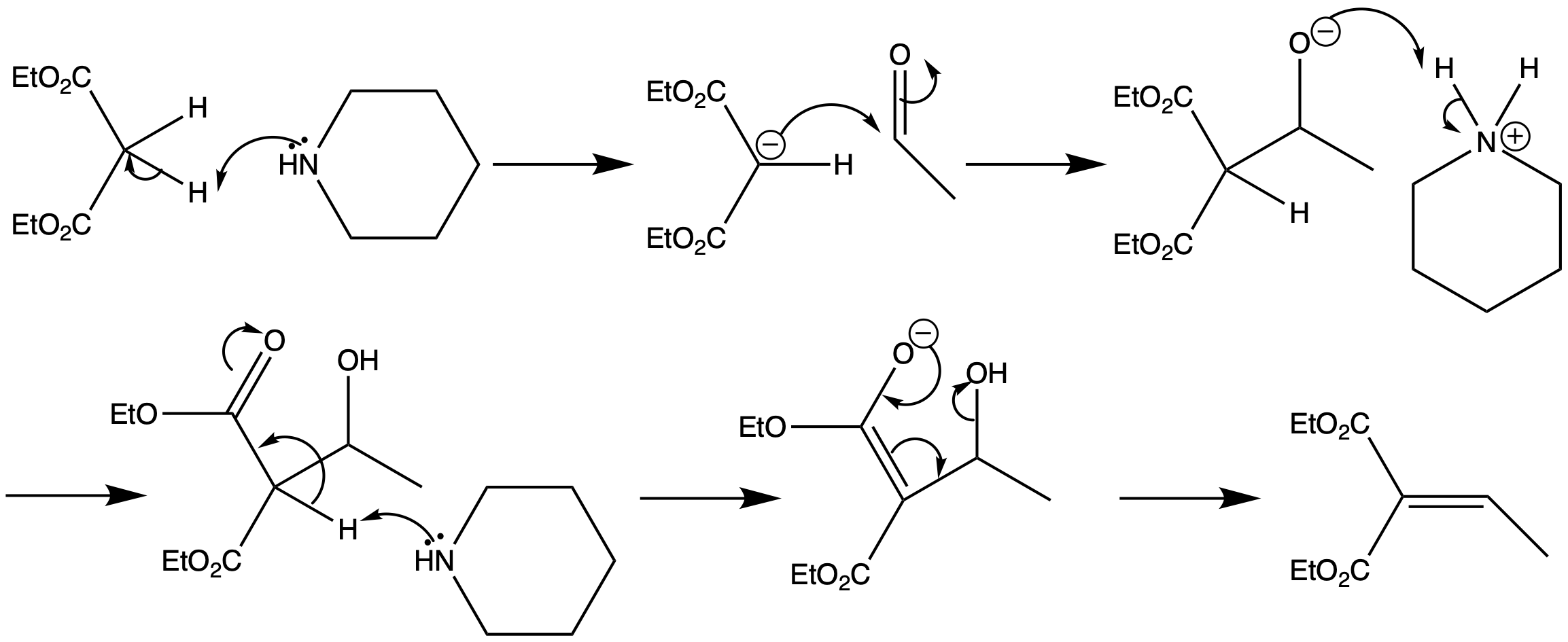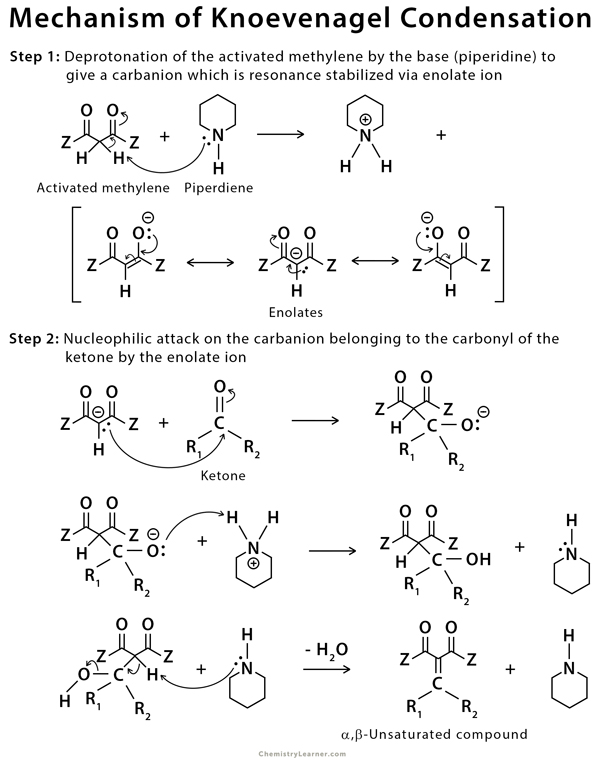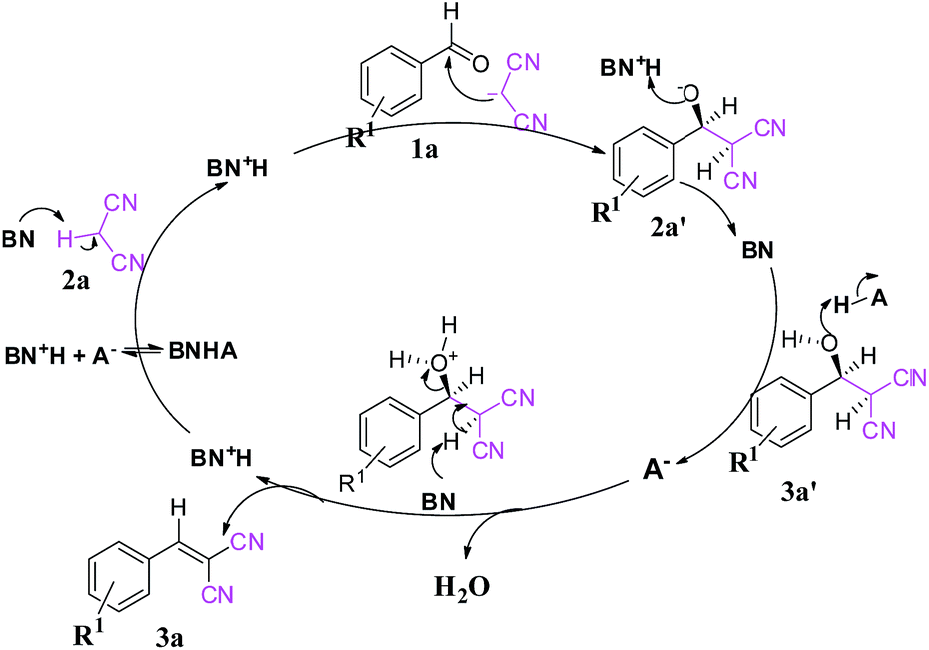
First report of the application of simple molecular complexes as organo-catalysts for Knoevenagel condensation - RSC Advances (RSC Publishing) DOI:10.1039/C5RA09036A

Contribution of Knoevenagel Condensation Products toward the Development of Anticancer Agents: An Updated Review - Tokala - 2022 - ChemMedChem - Wiley Online Library
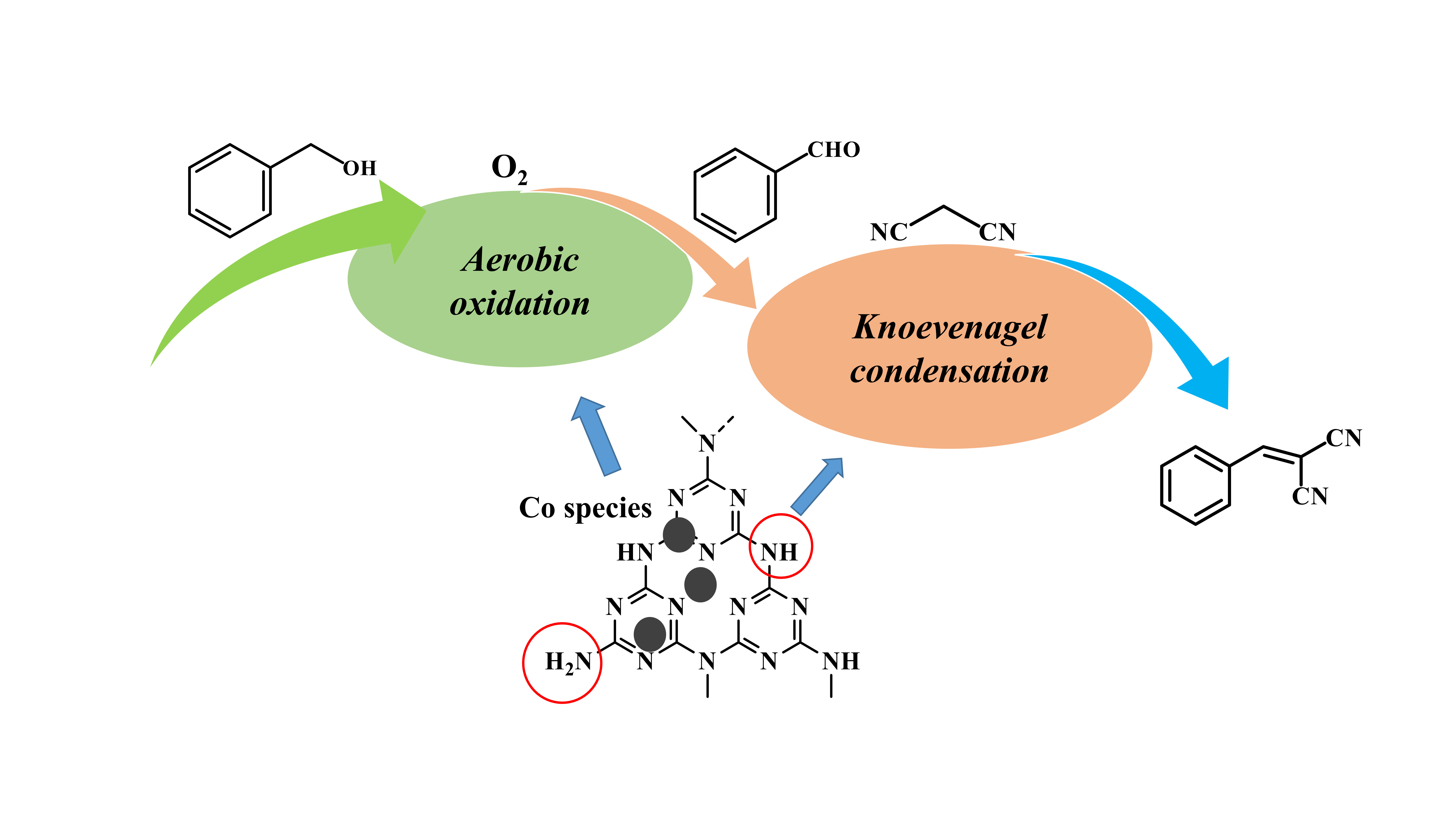
Catalysts | Free Full-Text | A Highly Efficient Bifunctional Catalyst CoOx/tri-g-C3N4 for One-Pot Aerobic Oxidation–Knoevenagel Condensation Reaction